 As heat is added and the amount of gas increases, the piston cannot move up. In a constant volume system, the volume of gas above the liquid cannot change ( Figure 3). We’ll explore this very important concept of enthalpy in greater detail later in the module. If we rearrange the first law of thermodynamics, ΔE = q p + w and substitute in w = -PΔV, we get: We’ll refer to the heat that flows in or out of a constant pressure system as q p. The expansion of the gas at a constant pressure does work on the surroundings. In a constant pressure system, the volume of gas on top of the liquid can change as more gas is produced from the evaporation of the liquid. #First law heat and work in thermodynamics calculator how to#In later classes, you will learn how to calculate just how efficient this conversion is. This partial conversion of heat into work is the basis of much of our modern society. The piston does work on the surroundings! Pistons used in our cars then move a crank shaft, the wheels turn, and the car or train or boat is pushed forward. If heat flows out of the system, q out, or work is done by the system, w by, its internal energy decreases, ΔE 0), and thus w = –PΔV < 0. The system expends energy ( w 0) when heat flows into the system heat is negative ( q 0) when work is done on the system work is negative ( w 0. As the physical change occurs and gas is generated, the bag inflates and the volume increases (ΔV > 0). The system includes everything inside the bag: CO 2(s), CO 2(g), and a small amount of air. If the solid CO 2 is placed in a sealed zip-lock bag and a book is placed on top of it, then work can be done by the system to lift the book as the CO 2(s) sublimates. One simple system is the sublimation of solid carbon dioxide to form gaseous carbon dioxide. If the volume instead decreases (compression, ΔV 0 and energy is added to the system. The negative sign is necessary to catalog energy transfer from the system’s point of view: w 0) and energy is transferred out of the system into the surroundings. A simplified view of work states that a change in volume must occur and is expressed from the system’s point of view with the equation: As the volume of the tire increases work is also done by the expanding tire as the bicycle will rise slightly. For example, we do work when we expel the air from a pump when inflating a bicycle tire. Formally, work is the process of causing matter to move over a distance. Work differs from heat by the way that collisions between molecules are harnessed, as discussed below for a piston to produce directed motion. We discussed heat in the previous section-it is the transfer of energy by myriad random collisions between molecules. Instead, energy transfer is characterized as work ( w) or heat ( q) between system and surroundings. The transfer of energy between substances is not categorized as kinetic or potential (which are properties of the molecules and not energy transfer). When thermal energy is lost, the kinetic energy of these motions decreases. The greater kinetic energy may be in the form of increased translations (travel or straight-line motions), vibrations, or rotations of the atoms or molecules. The internal energy of a substance is increased when the kinetic energy of its atoms or molecules is raised or its chemical potential energy is increased. 
The total energy is often called the internal energy (E), sometimes symbolized as U. 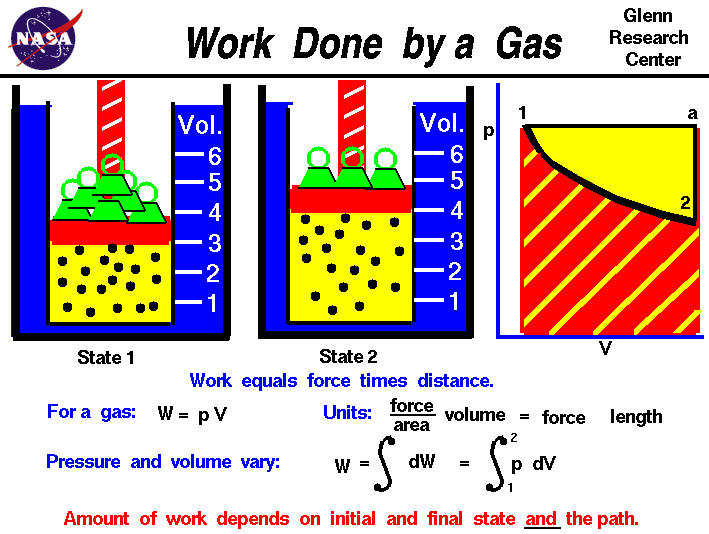
Substances act as reservoirs of energy by storing energy in the translations, vibrations, and rotations of the molecules (kinetic energy) and in chemical bonds (chemical potential energy).  
| Key Concepts and Summary | Key Equations | Glossary | First Law of Thermodynamics | First Law of Thermodynamics | Harnessing Heat: Work and the Movable Wall | Recognize the first law of thermodynamics as a statement of conservation of energy, with heat and work as two forms of energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
